|
1/7/2024 0 Comments Lattice energy equationAccording to this equation, stronger interactions occur between ions with larger charges and smaller radii. 
(1) so as to separate in the right-hand part of the equation the work of. Lattice energy depends on the strength of interactions between cations and anions in the lattice, which we can estimate using Coulomb's law: F (qq)/r. The lattice energy of an ionic compound is the energy change when one mole of ionicsolid is separated into its gaseous ions. (pyrophosphate) of formula M4P2O7(s) where M. The Born exponent is typically between 5 and 12. Crystal lattice energy and its calculation by means of ionic charges and radii. Abstract: Lattice energies and Gibbs energies of formation are determined for a series of alkaline diphosphate. The Born–Landé equation gives an idea to the lattice energy of a system. For the IB it is defined as the energy released when 1 mole. Lattice Energy or Lattice Enthalpy H L rm Delta HL HL, is the enthalpy associated with the formation of gaseous ions from the solid ionic substance. E = − N A M z + z − e 2 4 π ε 0 r 0 ( 1 − 1 n ) Calculated lattice energies Lattice enthalpy may be seen defined in two different ways, depending on the reference literature. In 1918 Max Born and Alfred Landé proposed that the lattice energy could be derived from the electrostatic potential of the ionic lattice and a repulsive potential energy term. These values are plugged into the Born-Haber equation: Lattice energy -146.2 - (+147.1) - 520 - (0.5158.739) - (-328) -564.7 kJ/mol. 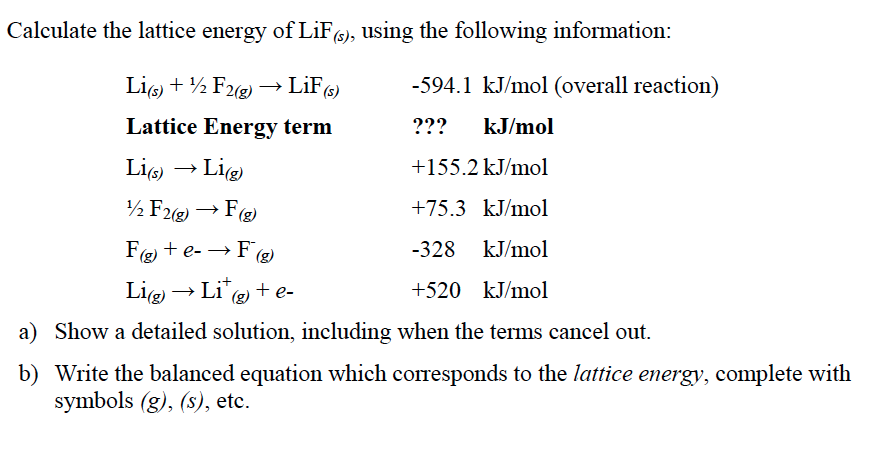
The Born–Landé equation is a means of calculating the lattice energy of a crystalline ionic compound. This article will show you the lattice energy equation, lattice energy formula, how to calculate it step by step, and give you examples of how the equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
